| +48 22 810 80 17 | biuro@mikrolab.waw.pl |
Microbiological testing of cosmetic products
Cosmetic products that hit the market must be safe for consumers. In order to ensure a high level of health protection for users, a cosmetic product should be manufactured in accordance with the requirements of Good Manufacturing Practice and meet the relevant legal requirements.
One of them is the need to perform microbiological tests resulting from the Regulation (EC) No. 1223/2009 of the European Parliament and of the Council of November 30, 2009, which has been in force since July 11, 2013. In accordance with the applicable regulations, tests of cosmetic products should be performed in reliable and reproducible way, which ensures that analyzes are carried out in accredited laboratories.
Microbiological purity tests of cosmetic products are carried out, among others: in order to control manufacturing conditions and check the quality of the finished product. The aim of the tests is to detect microorganisms in the product, including those that may be potentially pathogenic to consumers. The scope of the research includes: quantitative analyzes for the total number of aerobic microorganisms, the total number of yeasts and molds, and qualitative analyzes for the presence of specific, undesirable microorganisms in cosmetic products, i.e. Pseudomonas aeruginosa, Staphylococcus aureus, Candida albicans and Escherichia coli. The currently applicable methods for testing cosmetics are the PN-EN ISO standards, which contain guidelines for testing cosmetic samples. To be safe, a cosmetic product must meet the requirements of the PN-EN ISO 17516:2014-11 standard, which includes permissible limits for microbiological contamination.
For cosmetics intended for children under three years of age, used around the eyes or mucous membranes, the maximum acceptance limit for the total number of aerobic microorganisms (bacteria and fungi) is 2.0 x 10 2 cfu/g, and for other products - 2.0 x 10 3 cfu/h Both groups of cosmetic products must be free from specific microorganisms (Pseudomonas aeruginosa, Staphylococcus aureus, Candida albicans and Escherichia coli) in 1 g or ml of the product. The PN-EN ISO 17516:2014-11 standard refers to the standards according to which quantitative and qualitative tests of cosmetic products are performed.
The number of aerobic microorganisms is determined according to the PN-EN ISO 21149:2017-07 standard, while the number of yeasts and molds is determined according to PN-EN ISO 16212:2017-08. Tests for determining the presence of undesirable microorganisms in cosmetic products are performed according to the following standards:
- PN-EN ISO 22717:2016-01 – detection of the presence of Pseudomonas aeruginosa,
- PN-EN ISO 22718:2016-01 – detection of the presence of Staphylococcus aureus,
- PN-EN ISO 18416:2016-01 – detection of the presence of Candida albicans,
- PN-EN ISO 21150:2016-01 – detection of the presence of Escherichia coli.
These tests are performed in 1 g or ml of the product by multiplication in a broth medium and then inoculation on selective agar media for each specific microorganism. Before testing samples of cosmetic products according to PN-EN ISO standards, it is necessary to check the effectiveness of neutralization of the antimicrobial properties of the tested sample. The test is performed separately for each cosmetic mass. This is to verify the test conditions, i.e. to check whether given microorganisms can grow in the conditions in which the analysis is carried out. The standards indicate neutralizers that can be used, but sometimes another effective neutralizing medium must be selected for a given product. Tests for the presence of the indicated microorganisms are aimed at detecting pathogenic microorganisms in cosmetic products, the presence of which disqualifies a given product.
ISO standards additionally introduce detection of the presence of Escherichia coli, which aims to check whether production takes place in proper hygienic conditions. The presence of Escherichia coli in the product may indicate contamination of fecal origin. Testing the microbiological purity is important due to the fact that the vast majority of preparations used for care are susceptible to microbiological contamination. The source of infections, apart from improper production hygiene, are primarily raw materials. Cosmetics containing raw materials, such as water or natural raw materials, are most susceptible to microbiological infections. Water used for production may be a source of Pseudomonas bacteria, including Pseudomonas aeruginosa. However, natural raw materials include various types of plant extracts, crushed nut shells or other plant parts, as well as mineral substances that may be a source of diverse microflora.
Bacillus bacteria and mold fungi can often be found in natural raw materials. The use of contaminated products may pose a serious health risk. The presence of pathogenic bacteria Pseudomonas aeruginosa may cause various infections, e.g. conjunctivitis, corneal ulcers or urinary tract infections. Products containing Pseudomonas microorganisms, e.g. mascaras, may consequently lead to serious health complications, even damage to eyesight. The presence of other pathogenic microorganisms - Staphylococcus aureus may cause skin infections, inflammation of the throat, sinuses, nose, and urinary tract infections. These microorganisms are found in body creams, balms and mascaras, and a potential danger may arise from the use of such products on damaged skin or the surface of the eye. The presence of Candida albicans in cosmetic products, e.g. hand creams or products used around the eyes, may cause fungal infections of the skin, nails, as well as conjunctivitis or cornea. The assessment of microbiological purity is important both for the protection of the health of users of cosmetic preparations and for the quality of products offered on the market.
Microbiological contamination of cosmetics may reduce the quality of products by changing consistency, color or unpleasant smell, and the use of such a product may lead to allergic reactions, irritations or skin infections. A cosmetic manufacturer, wanting to ensure the high quality of its products offered on the market, should protect the cosmetic against possible secondary infection. Microbiological contamination may occur in a product not only at the production stage, but also during storage and use of a given preparation. Cosmetics susceptible to infections should be protected during the development of their formula by adding appropriate preservatives. This is particularly important in the case of products containing water and dosed in multi-dose packaging, to which the consumer can introduce an infection through contact of the cosmetic with microorganisms found on human skin. The task of preservatives is to protect the cosmetic product against the growth of microorganisms, including pathogenic microorganisms. The effectiveness of the preservatives used is checked by conducting a test, the so-called maintenance test, challenge test, also known as a load test. For cosmetic products, maintenance tests are carried out according to the PN-EN ISO 11930:2019-03 standard. The test involves a single introduction of a suspension of known density of specific microorganisms into the product under laboratory conditions: bacterial strains (Pseudomonas aeruginosa, Staphylococcus aureus and Escherichia coli), a strain of Candida albicans yeast, a strain of Aspergillus brasiliensis mold fungi, and leaving the inoculated product in appropriate conditions for a specified period of time. contact of microorganisms with the product. The infected products are then inoculated at specific intervals to determine the degree of microbial reduction under the influence of preservatives.
The applied preservative system is considered effective if, after a specified contact time, the number of microorganisms introduced into the product is reduced as required by the standard:
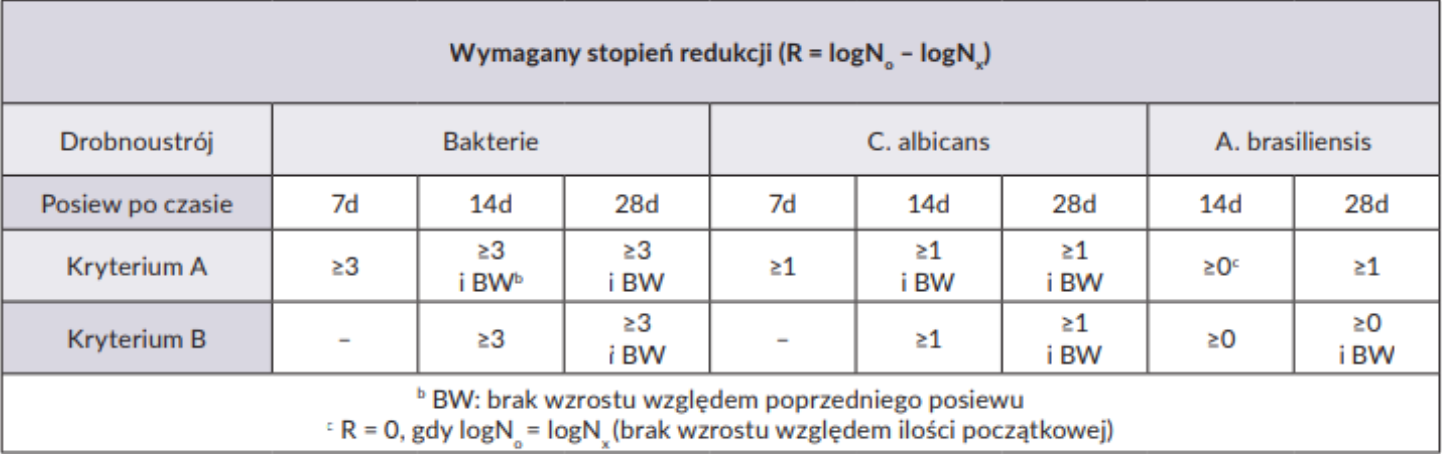
In addition to the strains indicated in the PN-EN ISO 11930:2019-03 standard, environmental (in-house) strains occurring in a given production area, representing possible contamination of the product during its production, can also be used.
Maintenance tests are carried out:
- in order to select the appropriate preservative at the stage of laboratory product development to confirm the proper operation of the preservatives used,
- in production series to confirm that the technological process does not affect the stability of the preservation system,
- to protect the product in the event of uncontrolled contamination during consumer use or storage,
- to confirm the stability of preservatives during the product shelf life,
- to select the appropriate concentration of a preservative that is both effective in protecting the product and safe for the user's health,
- for safety assessment in the presence of environmental strains causing microbiological problems.
In order for a cosmetic product to be introduced to the market, it is necessary to perform a preservation test, which aims to demonstrate proper protection of the cosmetic during its storage and use. Preservation tests allow you to assess whether a given product is properly protected against the development of undesirable microorganisms that may be a source of infection for consumers using the cosmetic or cause changes in the product that affect its quality. It is important that the use of preservatives is not intended to replace the principles of Good Manufacturing Practice, but to protect the product against unexpected contamination during production, storage or use. Not all cosmetic products must be subject to microbiological testing. Products with low water content, bath salts, aerosol cosmetics and, above all, those based on alcohol or containing acids are at the least risk of microbiological infection. The PN-EN ISO 29621:2017-04 standard presents guidelines for the assessment of products that can be classified as low microbiological risk. This group includes:
- products with pH ≤3.0, e.g. peelings,
- products with pH ≥10.0, e.g. hair straightening products,
- products containing ≥20%. alcohol, e.g. perfumes, tonics,
- products with low water activity ≤0.75 and high process temperature, e.g. lipsticks, lip balms, pressed powders,
- solvent-based products, e.g. nail polishes,
- oxidizing products, e.g. hair dyes.
The main goal of cosmetic product manufacturers should be to produce high-quality products that are safe for consumers. Ensuring the safety of products available on the market results from legal requirements imposed on producers, primarily Regulation (EC) No. 1223/2009 of the European Parliament and of the Council. This is important not only because of the production of a product that is safe to use and meets the microbiological quality requirements, but also to maintain a good image of the company. Production based on a high standard of hygiene, appropriate quality control of raw materials and finished products can protect the manufacturer from releasing products of inadequate quality to the market, which may consequently result in marketing and financial losses.
